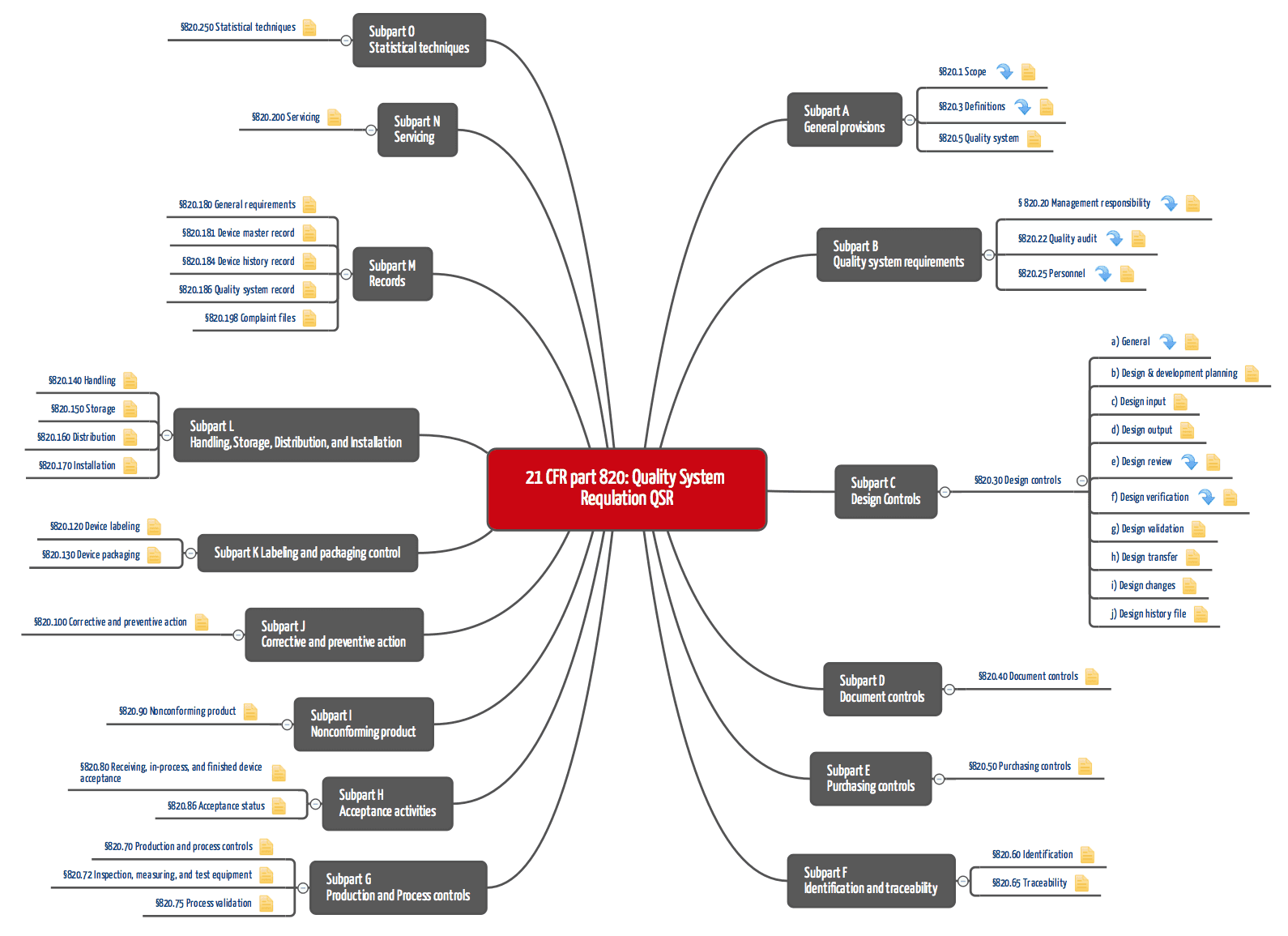
There are two major updates of this clause in ISO 13485:2016 when compared to the previous version. ISO 13485:2016 Clause 4.2 Documentation Requirements However, the interpretation and application of risk-based approaches is consistent with FDA expectation.Ģ. The FDA 21 CFR Part 820 does not explicitly define risk-based requirements for a quality system in the regulations. 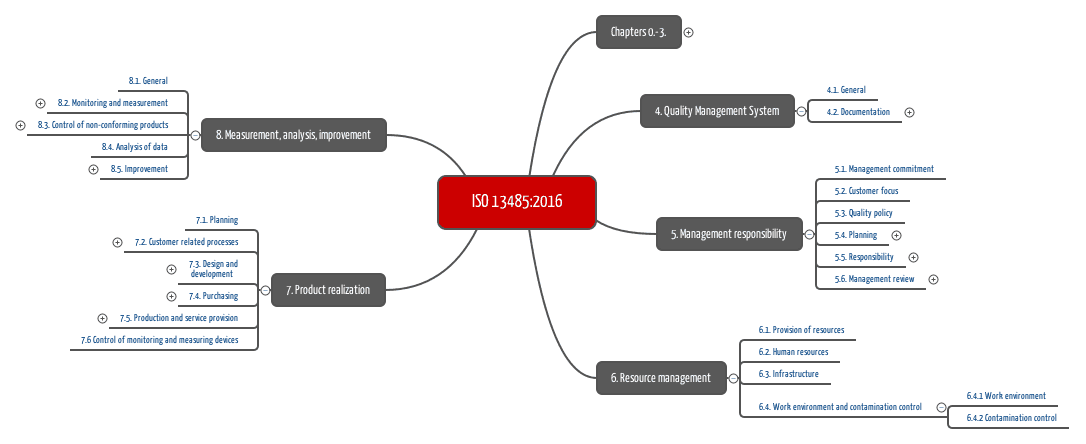
ISO 13485 now requires the application of a risk-based approach to establishing and maintaining a QMS. ISO 13485:2016 Clause 4 Quality Management System & 4.1 General Requirements With this short article, you have a comparison of how these changes relate to FDA 21 CFR Part 820.ġ. Compared to the earlier version of ISO 13485, eleven clauses have significantly changed in the 2016 edition of the ISO 13485.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
